Basics of X-Ray Crystallography
Basics of X-Rays
Q1. What are X-Rays?
Q2. Basics of X-Rays
Q3. Applications of X-Rays
A1. X-rays are a form of electromagnetic radiation, just like visible light. Electomagnetic radiation is a part of Electromagnetic Spectrum , this is comprised of a group radiation that are collectively called as electromagnetic radiation.
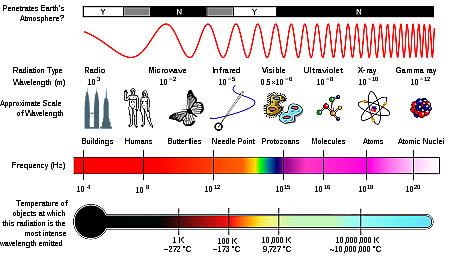
There are many different kinds of electromagnetic radiation, like the visible light that allows us to see colors, or the radio waves that transmit sound over large distances, or the even microwaves we use to cook our food. Infrared and ultraviolet light comes from the sun; infared is responsible for causing the global warming, and ultraviolet penetrates the atmosphere and causes skin cancer. Also, X-rays and Gamma are part of the electromagnetic spectrum, these rays are generated by much hotter and energetic objects like stars!
X-rays are most commonly used by your doctor or dentist to look at your bones and teeth. X-rays have a wavelength between 10 to 0.01 nanometers, They are shorter in wavelength than visible light rays.
In chemistry, one of the uses of X-rays are in X-ray crystallography. X-ray crystallography is a method of determining the arrangement of atoms within a crystal. X-rays strikes a crystal and scatters into many different directions, the way the X-rays scatter gives researchers an idea of the structure.
Q2. How are X-rays generated in the laboratory?
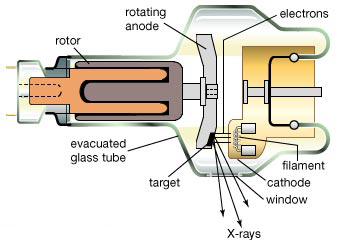
A2. In the laboratory, an X-ray is generated inside a vacuum tube. High energy electrons are emitted from the cathode (the positive end) and strike the anode (the negative end). In the figure below, a beam of high energy electrons are emitted by heating the cathode (usually copper), which then travel towards the rotating anode (usually tungsten) and upon striking the anode, come to a complete stop. The energy released on such a strike is released as X-rays, which then come through a window in a desired direction.
Q3. What are the various applications of X-rays?
A3. X-rays can penetrate less dense objects, but cannot pass through high-density objects. This makes X-ray a useful tool for looking at places which are otherwise opaque to human eyes. X-rays have applications in various aspects of our lives, like medicine or dentistry, orthopedics, Computer axial tomography (CAT scan), and in the laboratory. X-rays are excellent for nondestructive analysis (XRD, EDX, etc). In forensic labs it is used to determine the elements in any sample under investigation. In the transportion industry, X-rays are used to scan the luggage at the airports thus revealing the inner contents of any bag. X-rays are also used in various industries for quality control, quality assurance, routine maintenance, and monitoring of crucial parts.